Cancer Biology and Evolution Program Aims
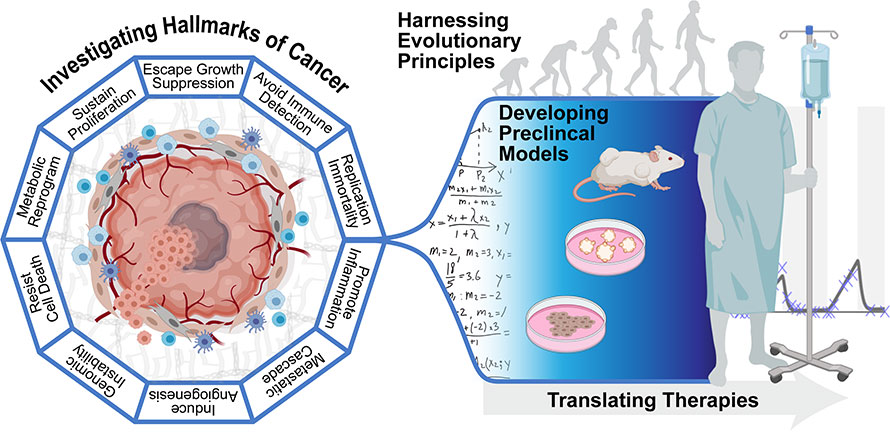
Graphic adapted from Hanahan and Weinberg, Cell (2011)
Aim 1: To define molecular and evolutionary networks that drive cancer development, progression, metastasis and resistance. The identification of molecular networks driving cancer are crucial to build an understanding of the disease, to model it in vivo, and to further design effective therapies. The goal of Aim 1 is to unravel the complexities of cancer development and progression and to identify biomarkers of tumor development, progression and response to therapy to further the goals of Aims 2 and 3.
Aim 2: To develop animal and mathematical models of human cancer development, progression, metastasis and therapeutic resistance. To effectively understand cancer development, progression, metastasis and therapeutic response and resistance, one of the primary goals of CBE is to generate in vivo and in silico models to understand the disease: initiation, progression, metastasis, and the tumor microenvironment. These models take the form of sophisticated genetically engineered mouse models (GEMMs) and mathematical models that account for a multitude of parameters during tumor evolution, growth, progression, metastasis and therapeutic responses. The goal of this Aim is to provide a platform to understand the evolution of cancer and metastasis for translational cancer research.
Aim 3: To translate basic discoveries into personalized, mathematically-informed therapies. A critical component of the CBE research paradigm is integration of mathematical models into treatment strategies. The term "personalized medicine" is often applied to strategies that match tumor treatment to specific predictive biomarkers measured from an individual patient. These approaches have prolonged patient survival times in certain cancer types. However, the reality is that the initial positive response is often followed by the evolution of drug resistance, treatment failure, and eventual patient death. Rather than ignoring this evolution, evolutionary-enlightened therapeutic approaches of CBE deliberately embrace the dynamics that govern the emergence of drug resistance as discovered through research in Aim 1, modeled in Aim 2, and further develop treatment strategies that exploit these dynamics in Aim 3. These include clinical trials to address the cancers that contribute to higher incidence and mortality (e.g., melanoma and lung) and disparities (e.g., prostate cancer) in our catchment area.
Cancer Biology and Evolution
