Fluorescent Imaging Could be ‘Game Changer’ in Breast Cancer Surgery
After a breast cancer diagnosis, many women face a daunting decision: choosing a mastectomy to remove the breast, or a lumpectomy to remove a tumor inside the breast.
For more than 90,000 women each year in the United States, the fear that some cancer will be left behind is too great, and they choose a mastectomy.
Those concerns are validated by the more than 80,000 women a year who must undergo a second surgery because the lumpectomy failed to remove all of the malignant tissue.
A new technology could change the dynamics of that frightening decision.
Surgeons have begun using a fluorescent cancer marker during surgery to help them identify microscopic cancer cells in real time, greatly reducing the need for repeat surgeries and improving patient outcomes.

Catherine Lee, MD, surgeon, Breast Oncology Program
“Fluorescent imaging systems are trying to bridge the gap between what we can see with the naked eye,” said Dr. Catherine Lee, a surgeon in the Breast Oncology Program at Moffitt Cancer Center. “If I take out what I think is a tumor and then use a fluorescent camera to light up what remains, it would be a game changer.”
JoAnn Runk was one hoping this technique would spare her from a second surgery and save her life. As an executive assistant in Moffitt’s Breast Oncology Program, she knew breast cancer was not uncommon. However, she never thought she would personally be affected until she was diagnosed in 2017.

JoAnn Runk was diagnosed with breast cancer in 2017.
“My eyes opened up a little more with my diagnosis,” she said. “It wasn’t just something that happened to other people.”
An avid supporter of Moffitt’s research mission, Runk was interested in joining a clinical trial and was eligible for one of the fluorescent imaging studies. Runk has a family member who a few years before needed a repeat surgery after unclear margins, and she hoped the trial could give her some peace of mind. She was the first Moffitt patient to enroll.
“The dye worked perfectly for me and surgeons got the clear margins they needed,” said Runk. “I didn’t have to come back for another surgery.”
The results from the phase 2 trial Runk was part of showed using a drug called pegloprastide, a fluorescent cancer marker, during surgery correctly identified cancer in up to 75% of patients who would have otherwise been candidates for a repeat surgery. The drug is administered by intravenous infusion up to a day before surgery, and after surgeons remove the tumor, they use a near-infrared camera to identify any cancer left behind.
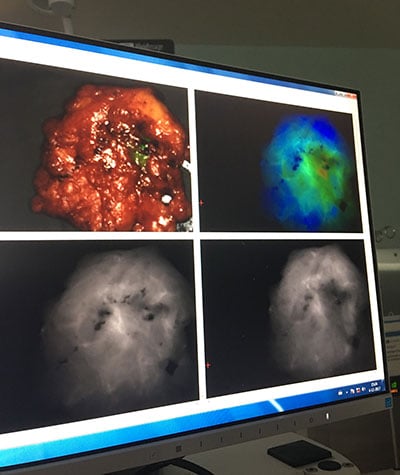
After surgeons remove the tumor, they use a near-infrared camera to identify any cancer left behind.
On average, 20% to 40% of patients who receive an original lumpectomy require a second surgery. However, only 6% of the 32 women on the trial needed a repeat surgery.
“These cancer-targeting agents have the potential to radically alter the practice of all cancer surgery, not just for breast cancer, and we are very excited to have this technology available to our patients,” said Lee.
Runk underwent radiation after her surgery and today is cancer free. She says she is grateful she had the opportunity to participate in the study, regardless of the results.
“In the beginning I had no proof that it would work, but the fact that it did was reassuring that this has a lot of potential to help others,” she said. “I am really glad I was able to do my part to help research in my small little way.”
Moffitt is opening a second fluorescent imaging trial this month that will test a different fluorescent marker and camera system.