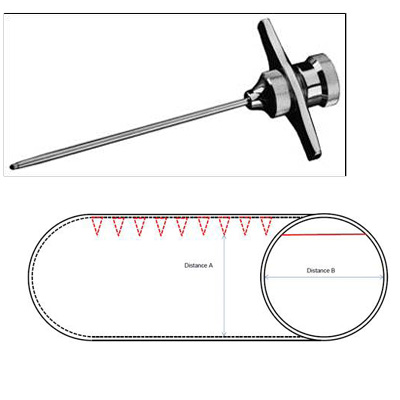
18MB047 Med Device: Biopsy Needle with Internal Ridges that Lead to a Higher Tissue Specimen Retrieval Rate
Biopsy needles are commonly used in hospital diagnostic procedures. Only about 40% of biopsies provide a sufficient sample using current instruments. Moffitt’s propriety designed biopsy needle provides more adherence between the biopsy core sample and the needle, and thus a higher retrieval rate for the tissue specimen. A prototype of the device was developed and tested in a pilot study and showed doubled bone biopsy retrieval rates compared to traditional biopsy needles.
COMMERCIAL OPPORTUNITY
- Core needle biopsies are a common way to take tissue samples from patients for laboratory testing. A doctor may use a needle biopsy to help diagnose a medical condition or to rule out a medical condition. For example, the needle biopsy may be used for a mass or lump to determine if it is a cyst, an infection, a benign tumor or a cancer.
- Regarding the number of core needle biopsies performed per year, more than one million women have breast biopsies each year in the United States, and about 20 percent of these biopsies yield a diagnosis of breast cancer (www.effectivehealthcare.ahrq.gov/breast-biopsy).
- A box of 10 Cardinal Tru-Cut Biopsy Needles (14G x 6”) has a retail price of $600, suggesting an annual market size of more than $60M just for breast biopsies alone.
TECHNOLOGY
The biopsy needle has a series of internal ridges that that result in a biopsy core sample D-like shape. Upon twisting the biopsy needle at least 90 degrees, the ridges can help capture the sample and retain it in the biopsy needle until the sample is properly released from the needle and stored. Traditional bone biopsy procedures have a success rate of only 20-40%. Unsuccessful biopsy retrieval leads to additional procedures which increases the time, cost and risk of patients. This device significantly increases the probability of a successful biopsy procedure while also ensuring more sample collection thereby improving diagnosis.
PUBLICATION/PATENT
PCT application filed in February 2020 for Dr. Nam Tran.
The Innovation Office
InnovationMarketing@Moffitt.org
(813) 745-6828

