Clinical Perspectives
216 Stories Found

Researchers Investigating TIL Therapy Feasibility in Head and Neck Squamous Cell Cancer

How a CRC Enhances the Patient’s Journey at Moffitt
FDA Approves CARVYKTI® and ABECMA for Early Treatment of Adult Patients with Relapsed or Refractory Multiple Myeloma
Advancements in Non-Muscle Invasive Bladder Cancer Treatment

Pioneering Advances in Upper Extremity Oncologic Reconstruction and Endoprosthetic Reverse Shoulder Technology
Advancements in Pancreatic Cancer Vaccines: A New Frontier in Immunotherapy Treatment
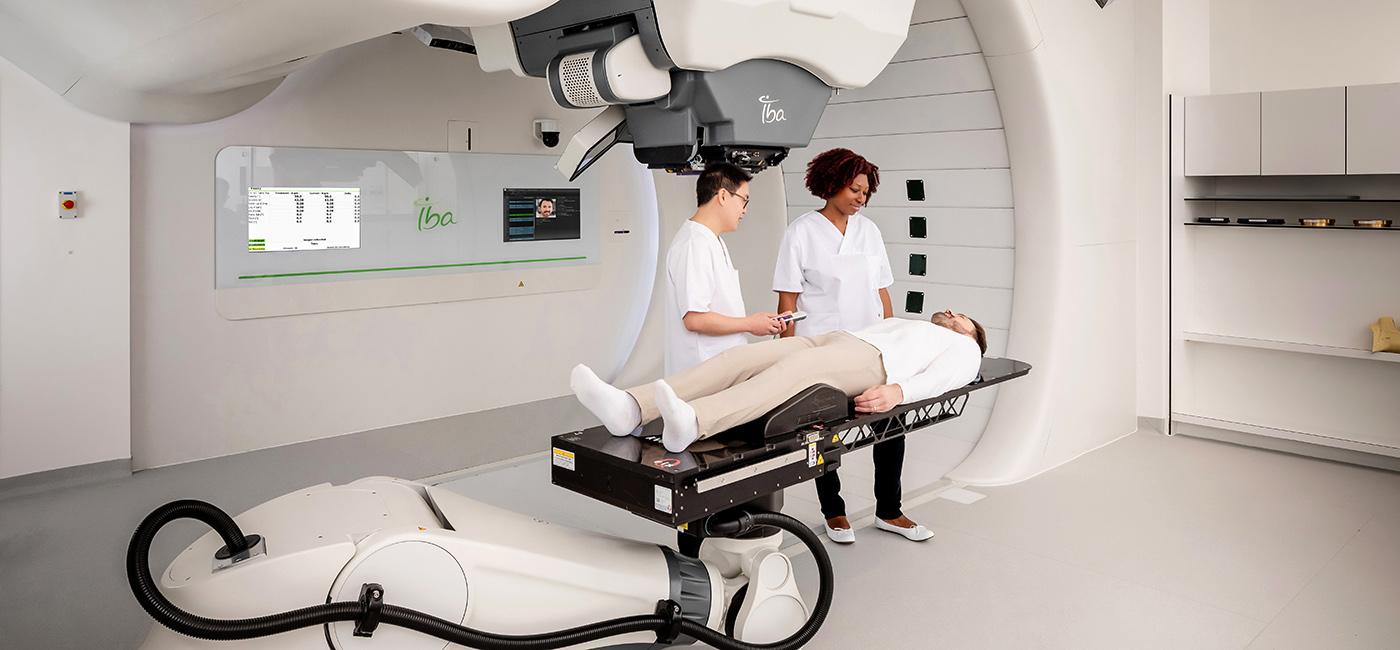
FAQ: Revolutionizing Radiation Treatment with Proton Therapy

Revolutionizing Thyroid Surgeries with Autofluorescence Technology
Revolutionizing Melanoma Treatment: FDA Approval for TILs Therapy Marks a Turning Point

Navigating New Frontiers: Far Medial Technique in Minimally Invasive Brain Tumor Surgery
Moffitt's CARDIO-CATCH Clinic for Clonal Hematopoiesis
Leaders in the Use of Navigated Bone Tumor Resections and Custom Implant Reconstruction

Advancements in Robotic Surgery for Radical Stump Trachelectomy
One of Only Five Multidisciplinary Cutaneous Lymphoma Clinics in U.S. is at Moffitt

Your Vote Counts! U.S. News Physician Survey is Underway
